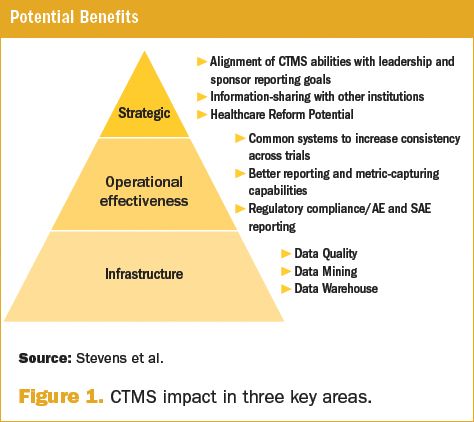
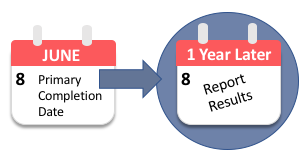
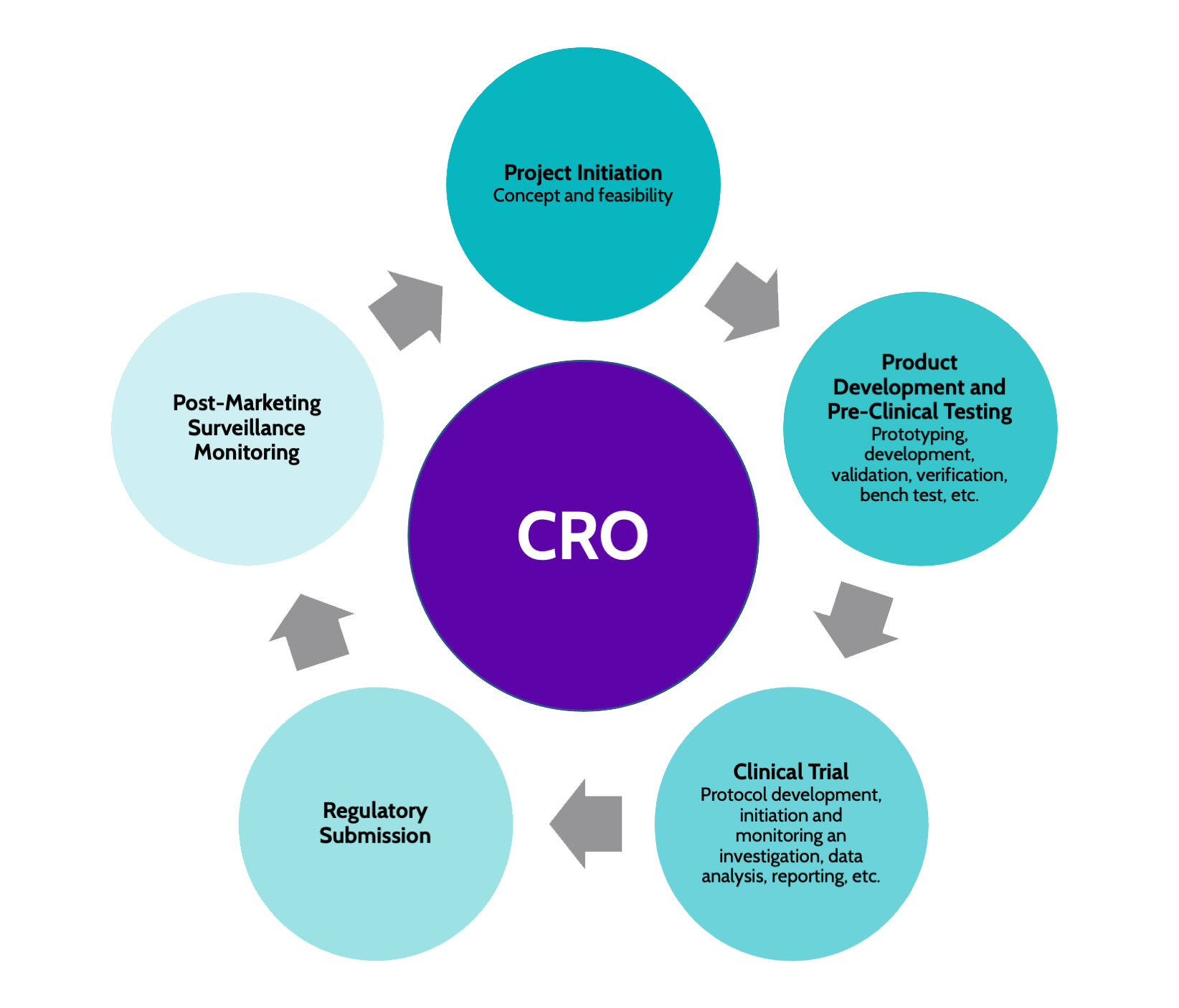
Safety Reporting Overload in Clinical Trials: FDA and Site Perspectives on Overreporting of Adverse Events | CenterWatch

Months and Severity Score (MOSES) in a Phase III trial (PARCER): A new comprehensive method for reporting adverse events in oncology clinical trials - eClinicalMedicine
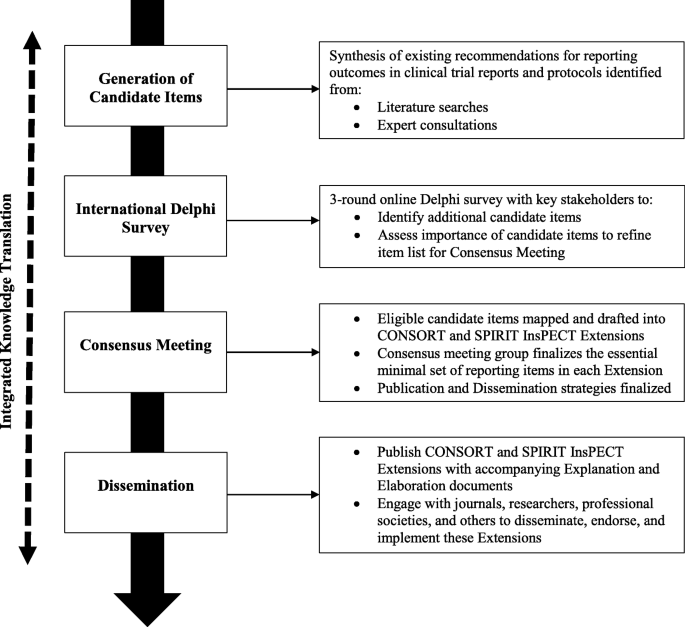
Improving outcome reporting in clinical trial reports and protocols: study protocol for the Instrument for reporting Planned Endpoints in Clinical Trials (InsPECT) | Trials | Full Text
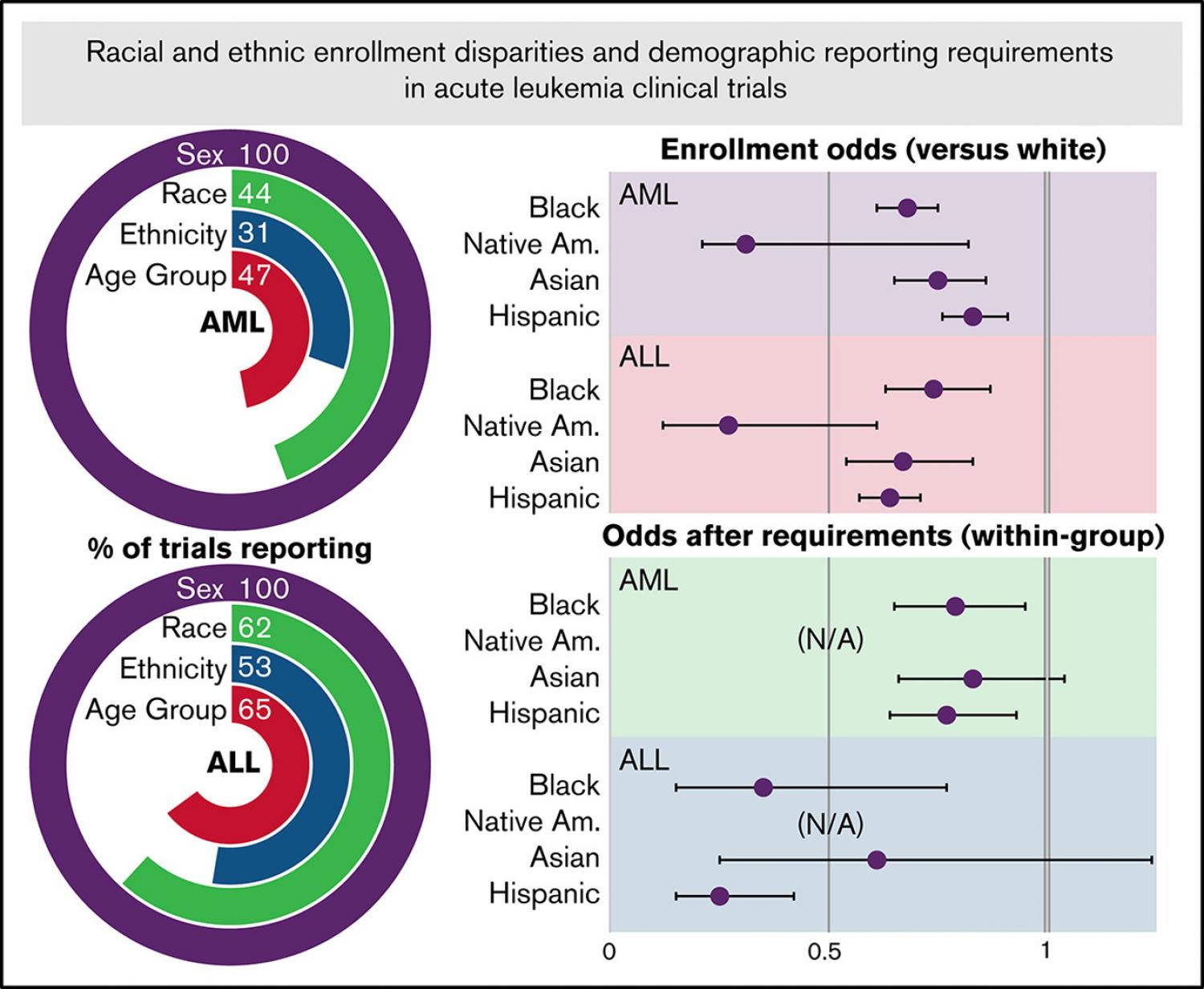
Racial and ethnic enrollment disparities and demographic reporting requirements in acute leukemia clinical trials | Sustainable Development Goals - Resource Centre

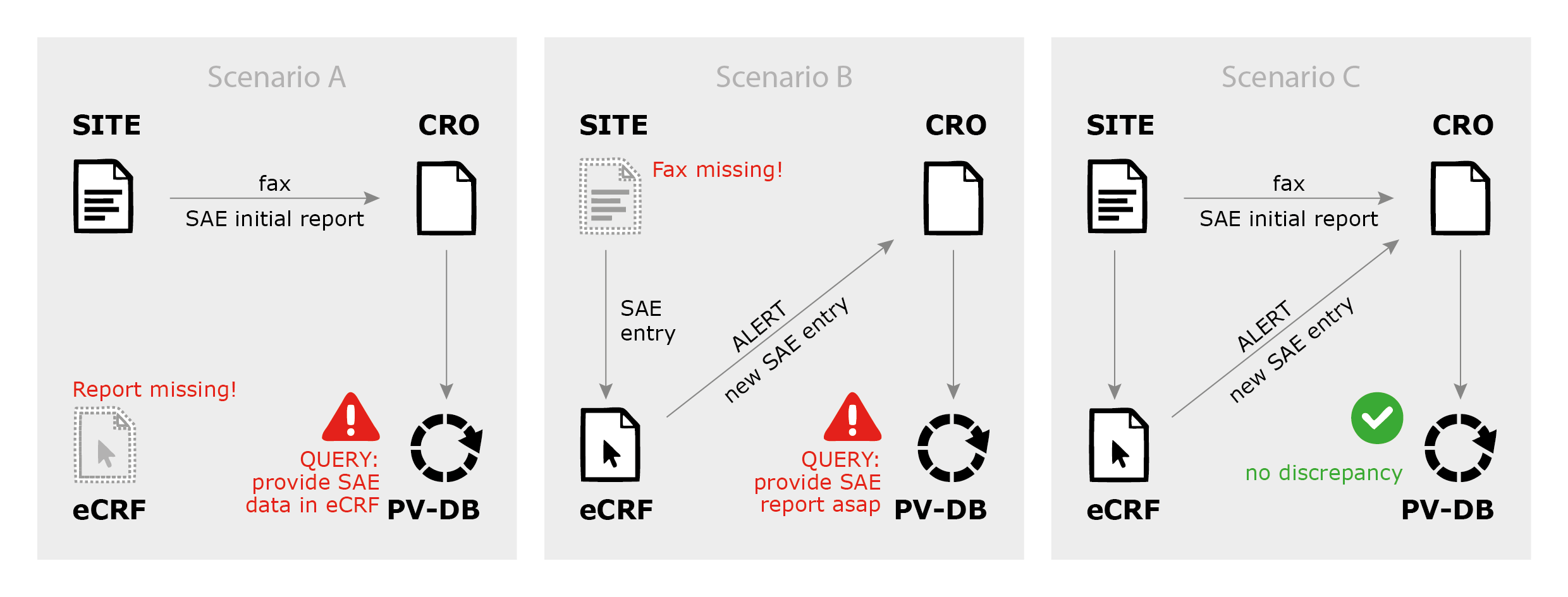
Process of reporting serious adverse events (SAE) during a regulatory... | Download Scientific Diagram